AF is the most common arrhythmia worldwide.1 It decreases quality of life and increases mortality in affected individuals.2,3 Recent studies have demonstrated that ablation therapy for rhythm control achieves a better clinical outcome than rate control therapy.4 However, the high recurrence rate of AF has halted the clinical impact of ablation therapy.
The risk factors for AF recurrence after ablation remain to be determined. Obesity has been reported as an independent risk factor for incident AF.5 A meta-analysis of western and Asian patients revealed that obesity is a significant risk factor for AF recurrence after radiofrequency ablation but that being overweight is not.6 However, the impact of increased BMI on cardiovascular disease may differ among races. Epidemiological studies have revealed that the distribution of BMI differs significantly among countries. For example, people in Asian countries have lower BMI values, and it has been suggested that a BMI of ≥25 kg/m2 in Asian people is similar to a BMI of ≥30 kg/m2 in white people.7 Although Asian people were included in the previous meta-analysis, the number was small, and the effects of ethnic differences were not assessed.6 Thus, the impact of BMI on AF recurrence after ablation in Japanese patients remains to be clarified.
There is a variety of ablation methods available, including radiofrequency ablation, cryoablation, laser balloons, hot balloons and pulse-field ablation. It has been reported that cryoablation is non-inferior to radiofrequency ablation.8 Specifically, cryoablation is more effective in reducing AF recurrence than radiofrequency ablation.9 Nevertheless, it is unclear whether BMI influences the likelihood of AF recurrence after cryoablation or radiofrequency ablation.
This study aimed to determine the impact of BMI on AF recurrence in Japanese patients and to assess the relationship between the impact of BMI and the ablation method. We prospectively examined AF recurrence in Japanese patients who underwent radiofrequency ablation or cryoablation.
Methods
Patients
We enrolled consecutive Japanese patients with non-valvular paroxysmal or persistent AF who underwent first-time single-catheter ablation from March 2013 to December 2019 at Tottori University Hospital, Japan. They were followed prospectively to detect AF recurrence after the procedure. We defined patients with a BMI of ≥25 kg/m2 as the overweight/obese group.
Paroxysmal AF was defined as self-terminating AF lasting for up to 7 days or AF episodes that exhibited cardioversion within 7 days. Persistent AF was defined as AF lasting between 7 days and 1 year or AF episodes that terminated either by cardioversion with drugs or by direct current cardioversion. The AF type and burden were determined using clinical reviews at least once a year, 12-lead ECG, device interrogation and 24-hour or 7-day Holter monitoring. Although there is no consensus on the duration of the blanking period, we defined the blanking period in accordance with current guidelines.10 The CHA2DS2-VASc score comprised congestive heart failure, hypertension, age ≥75 years, diabetes, stroke/transient ischaemic attack, vascular disease, age 65–74 years and female sex.11 We evaluated the body surface area using the formula of Du Bois [body weight (kg)0.425 × height (cm)0.725 × 0.007184].12 We calculated alcohol consumption of 20 units weekly as ethanol according to the HAS-BLED score.13 All laboratory measurements were assessed on admission (from 1 to 3 days before ablation procedures).
Recurrences of AF during the first 90 days after the ablation were not counted in the determination of the primary endpoints, and still included in the study.8
Study Protocol
The study protocol was approved by the institutional review board of our institution and written informed consent was obtained from each patient before the procedure. This investigation conformed to the principles outlined in the Declaration of Helsinki.
Intracardiac echocardiography-guided transseptal puncture was performed using a Brockenbrough needle. Immediately after transseptal puncture and pre-ablation blood sample collection from the left atrium (LA), an IV heparin bolus (150 IU/kg body weight) was administered, followed by continuous infusion to maintain a minimum target activated clotting time of >300 ms. LA sheaths were flushed continuously with heparinised saline at a steady rate of approximately 30 ml/h, and additional fluid replacement was initiated in cases of hypotonia throughout the procedure.
In the radiofrequency ablation procedure, pulmonary vein isolation (PVI) was achieved using a focal ‘point-by-point’ catheter approach, delivering radiofrequency energy to the cardiac tissue. A 3.5 mm irrigated-tip ablation catheter (Thermocool, Biosense Webster) was advanced into the LA to achieve bilateral circumferential pulmonary vein isolation with the endpoints of bidirectional conduction block between the LA and pulmonary vein using electroanatomical mapping (CARTO3, Biosense Webster) and fluoroscopy guidance. Details of the radiofrequency ablation procedure are described in our previous study, and cryoablation was performed as previously reported.14,15 PVI was performed with a single-balloon technique using a second-generation cryoballoon (CB; Arctic Front Advance, Medtronic). A 28 mm CB catheter was used in all patients. A spiral mapping catheter (Achieve; Medtronic) was used to advance the CB and to map the pulmonary vein (PV) potentials. Complete sealing at the antral aspect of the PV was confirmed via injection of contrast medium. To avoid phrenic nerve injury, diaphragmatic compound motor action potentials were monitored with phrenic nerve pacing during each CB application.
The procedural endpoints were defined as the establishment of bidirectional PV–LA block, which was verified via a circular mapping catheter. If electrical isolation was not achieved after CB applications (180 seconds for each application) on each vein, additional touch-up ablation was performed using conventional radiofrequency. Both an entrance block (i.e. the elimination of all PV potentials) and exit block (i.e. noncapture of the LA during placement of a circular catheter) were confirmed as endpoints following a 30 mg bolus injection of adenosine triphosphate, which unmasked any dormant PV conduction. After PVI, a bidirectional conduction block line was created at the cavotricuspid isthmus (CTI) in all patients, but the CTI was incomplete in five patients. The ablation procedure was performed under conscious sedation with continuous blood pressure and oxygen saturation monitoring. The decision of anti-arrhythmic drugs handled post-ablation was made by each attending physician.
The transthoracic echocardiography was performed from 1 to 30 days before catheter ablation. Echocardiographic data were measured according to the recommendation of the American Society of Echocardiography, which our previous studies followed.16
Statistical Analysis
Categorical data are presented as frequency (%) and were compared with Fisher’s exact test. Continuous data are presented as mean (SD) or as median (interquartile range) for skewed distributions. Normality was tested with the Kolmogorov-Smirnov test. Normally distributed continuous variables were compared using the independent Student’s t-test, and skewed data were compared using the non-parametric Mann-Whitney U-test. The Student’s t-test, analysis of variance and ANCOVA were used to compare the means between groups.
Analyses were performed after natural logarithmic transformation (ln) of skewed biomarkers. Freedom from AF was reported as crude event rates and by means of a time-to-event analysis using the Kaplan–Meier method. Variables with p<0.10 in the group comparison were further evaluated by the Cox proportional hazards regression analyses to determine their associations with the recurrence of AF. The variables with p<0.10 in the univariate Cox proportional hazards regression analysis were further evaluated by the multivariate Cox proportional hazards regression analysis. A trend analysis was performed using the Cochran-Armitage test and the Jonckheere-Terpstra test. All statistical analyses were performed using R (version 3.4.0; R Foundation for Statistical Computing), and p<0.05 was considered statistically significant.
Results
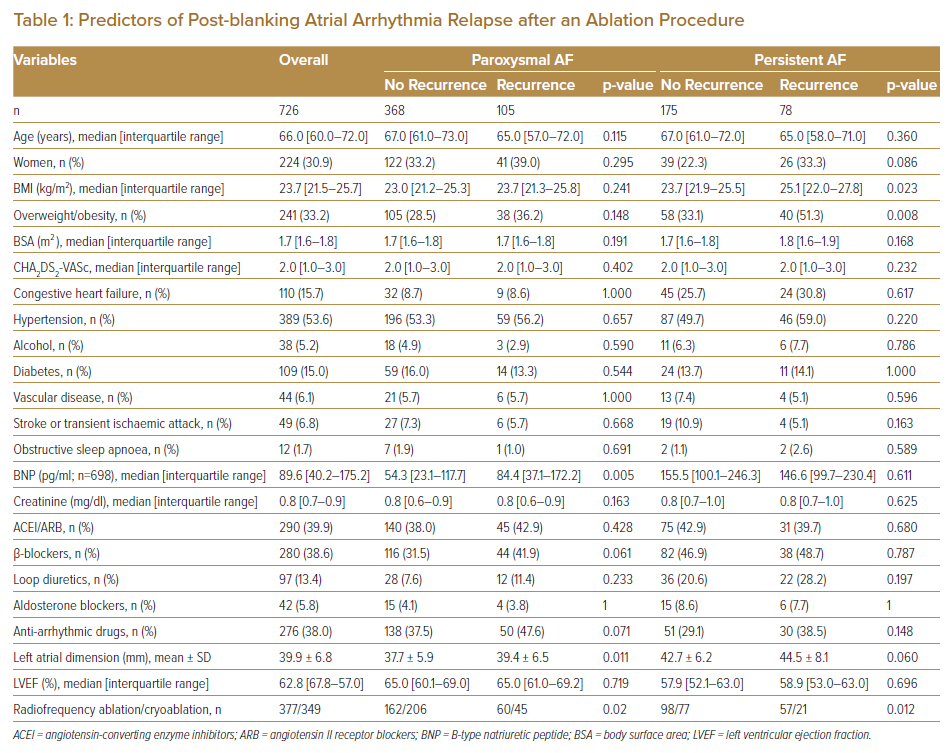
In total, 726 patients with AF (65.2% with paroxysmal AF and 34.8% with persistent AF) were included in this study (Supplementary Material Figure 1). The baseline characteristics of the patients are summarised in Supplementary Material Table 1. Most patients (69.1%) were men, and the median age of patients was 66.0 years [range 60.0–72.0]. The median follow-up duration for censored cases was 506.4 days [range 362.0–730.0]. Of all patients, 241 were classified as overweight/obese (BMI ≥25 kg/m2), and they had a significantly larger LA dimension (p<0.001). The median LA dimension was 39.8 mm and the median B-type natriuretic peptide was 89.6 pg/ml.
AF recurred in 183 patients (25.2%; 105 with paroxysmal AF and 78 with persistent AF). At baseline, no significant differences were noted in sex, age or left ventricular ejection fraction between patients with and without AF recurrence (Table 1). Table 1 shows the medication at the first day of hospitalisation. There were significant differences in the type of AF (paroxysmal versus persistent) and LA dimension between patients with and without AF recurrence. Median BMI was significantly higher in those with AF recurrence than in those without AF recurrence (23.9 kg/m2 [range 21.6–26.9] versus 23.2 kg/m2 [range 21.3–25.4], respectively; p=0.011). Moreover, the prevalence of patients treated with radiofrequency ablation was higher in patients with AF recurrence than in those without AF recurrence (p<0.001). The multivariate Cox proportional hazards regression analysis identified overweight/obesity (BMI ≥25 kg/m2) as an independent predictor of AF recurrence (adjusted HR 1.40, 95% CI [1.00–1.94]; p=0.049) (Supplementary Material Tables 2 and 3).
Subgroup Analysis of Risks of the Recurrence of AF for BMI
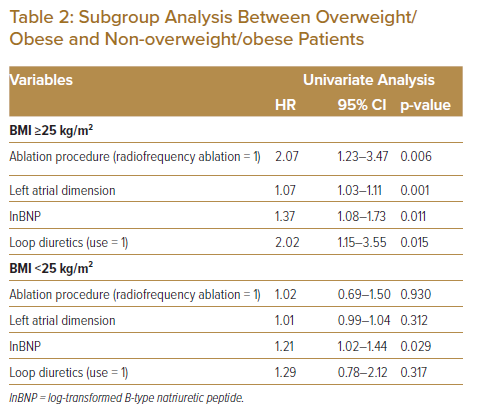
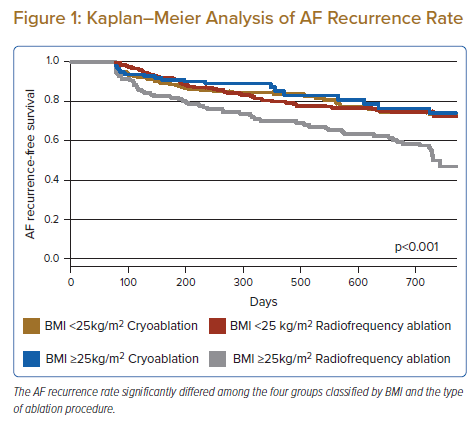
Based on the results above, we performed a subgroup analysis between the overweight/obese and non-overweight/obese groups. Fisher’s exact test showed that the AF recurrence rate increased significantly with BMI (non-overweight/obese: 21.6% versus overweight/obese: 32.4%; p<0.001). The AF recurrence rate in the overweight/obese group was significantly lower in the cryoablation group than in the radiofrequency ablation group (18.0% versus 44.6%, respectively; p<0.001). The multivariate Cox proportional hazards regression analysis identified larger LA dimension and radiofrequency ablation as independent predictors of AF recurrence in the overweight/obese group (radiofrequency ablation: adjusted HR 1.86; 95% CI [1.07–3.23]; p=0.03). The ablation procedure had no significant impact on AF recurrence in the non-overweight/obese group (Table 2). We stratified patients into four groups based on BMI and ablation procedure: BMI <25 kg/m2 and cryoablation; BMI <25 kg/m2 and radiofrequency ablation; BMI ≥25 kg/m2 and cryoablation; and BMI ≥25 kg/m2 and radiofrequency ablation. The AF recurrence rate significantly differed among the four groups according to the Kaplan–Meier analysis (log-rank test, p<0.001) (Figure 1).
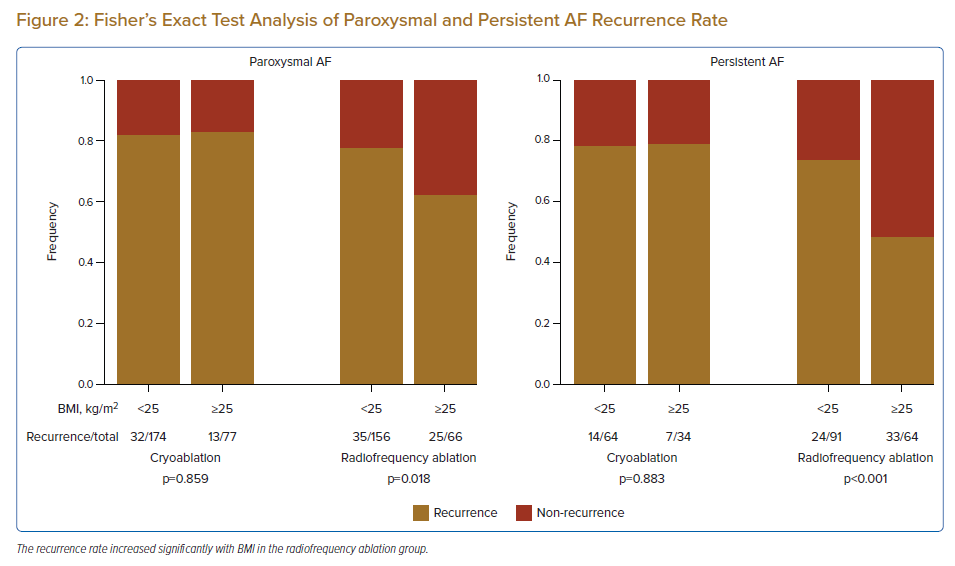
We further analysed the impact of AF type on AF recurrence. Fisher’s exact test showed that the recurrence rate increased significantly with BMI in the radiofrequency ablation group (normal BMI: 22.4% versus overweight/obese BMI: 37.9%; p=0.018) but not in the cryoablation group (normal BMI: 18.4% versus overweight/obese BMI: 16.9%; p=0.859) in patients with paroxysmal AF (Figure 2). Similarly, for patients with persistent AF, the recurrence rate increased significantly with BMI in the radiofrequency ablation group (normal BMI: 26.8% versus overweight/obese BMI: 51.6%; p<0.001) but not in the cryoablation group (normal BMI: 21.9% versus overweight/obese BMI: 20.6%; p=0.883). The Cochran-Armitage trend test showed similar results.
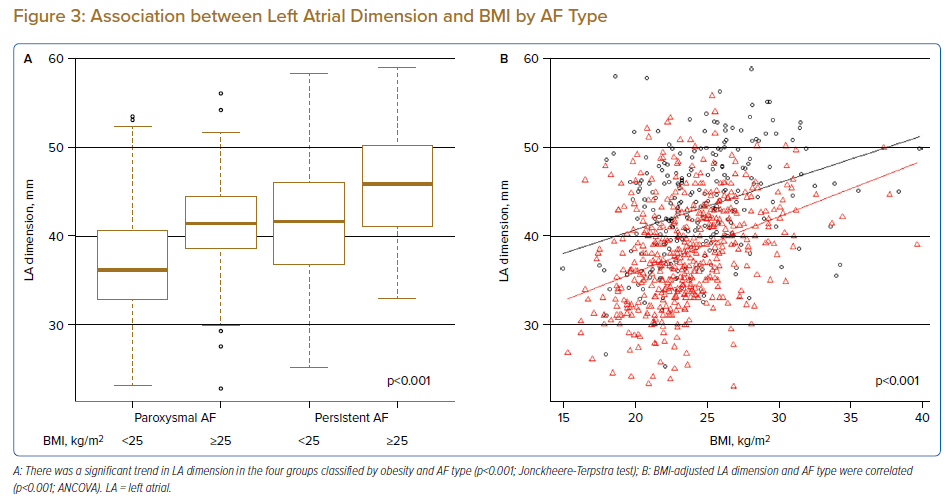
Association Between Left Atrial Dimension and BMI
Because LA dimension was an independent predictor of AF recurrence in the overweight/obese group, we examined the association between LA dimension and BMI in patients with paroxysmal versus persistent AF. The Jonckheere-Terpstra test showed that LA dimension increased in association with the increase in BMI and persistent AF (p<0.001; Figure 3A). Following the inclusion of AF type as an indicator variable and adjustment for baseline BMI, ANCOVA showed that LA dimension was higher in the persistent AF group than in the paroxysmal AF group (p<0.001; Figure 3B).
Safety of Ablation Procedures
Major complications occurred in 22 patients in the cryoablation group and in 13 patients in the radiofrequency ablation group. There was no significant difference in the occurrence of complications with ablation between normal weight and overweight/obese patients. As in previous studies, cryoablation was more frequently associated with diaphragmatic neuropathy, but all patients improved at 1-month post-ablation.8 A full list of postprocedural adverse events is provided in Supplementary Material Table 4.
Discussion
The aim of this study was to investigate the effect of BMI and type of ablation procedure on AF recurrence after ablation. Our results demonstrate that BMI was significantly higher in patients who presented with AF recurrence. We specifically identified that the AF recurrence rate of patients who underwent radiofrequency ablation, but not those who underwent cryoablation, increased with BMI. Patients in the overweight/obese group had a significantly larger LA dimension, and LA dimension enlarged synergistically with persistent AF and increasing BMI.
Although some meta-analyses have suggested that AF recurs more frequently in patients with a higher BMI after ablation, many subjects in these meta-analyses had a BMI of ≥25 kg/m2, and both western and Asian patients were included in the analysis.6,17 The studies in western patients reported obesity (BMI ≥30 kg/m2), but not overweight (BMI 25–30 kg/m2), as a risk factor for AF recurrence after ablation.18,19 In our study, the risk of post-ablation AF recurrence increased in Japanese patients with a BMI ≥25 kg/m2. BMI is a typical criterion to assess obesity, but it is influenced by race. It has been reported that Asian people have relatively lower BMI values compared with white people.7 Previous studies have indicated that the cut-off value for BMI as a risk factor for AF recurrence after ablation should depend on race.6,7
In the FIRE AND ICE trial, cryoablation was non-inferior to radiofrequency ablation with respect to their efficacy in treating paroxysmal AF.8 However, in recent years, cryoablation has been reported to reduce the incidence of paroxysmal AF recurrence compared with radiofrequency ablation.9 Another study has also shown that cryoablation is less operator-dependent and more reproducible than radiofrequency ablation in patients with paroxysmal AF.20 The pathological difference caused by ablation procedures may affect the AF recurrence rate after ablation. Reportedly, the LA–PV lesion caused by cryoablation is transmural, and the width of the lesion is 6 mm, which is greater than that of the lesion caused by radiofrequency ablation.21 Wider lesions caused by ablation may reduce re-conduction after ablation. Lesions caused by cryoablation exhibit less endocardial bleeding, are more clearly delineated and are more uniformly replaced with fibrous tissue. All these characteristics are expected to result in a low arrhythmogenic risk.21
The effect of different ablation procedures on AF recurrence in patients with obesity has not been well studied. Because LA dimension is an independent risk factor for recurrence in patients classified as overweight/obese, we analysed the relationship between BMI and LA dimension (Figure 3). LA size has been reported to be enlarged with persistence of AF and increasing body weight.22,23 In our study, LA dimension and BMI were correlated and LA dimension enlarged synergistically with persistent AF and increasing BMI. With an increase in BMI from 25 to 30 kg/m2, LA dimension increased by approximately 5 mm. With LA enlargement, radiofrequency ablation can increase the possibility of re-conduction because of an increase in the number of ablation points. This may at least partly explain the higher recurrence rate after radiofrequency ablation in the overweight/obese group in this study and in previous studies.
Obesity has been reported to increase the percentage of epicardial adipose tissue (EAT), and excessive pericardial fat is associated not only with a large LA volume and increased AF severity, but also with poor outcomes after radiofrequency ablation for AF.24–27 New insights may be gained by examining the impact of EAT from the perspective of the ablation procedure.
Clinical Implications
The current study, in line with the findings reported by Okumura et al., showed that overweight/obesity was associated with a higher rate of AF recurrence in Japanese patients after catheter ablation, suggesting that racial differences in BMI should be taken into account in evaluating the risk of post-ablation recurrence of AF.28 On the other hand, the subgroup analysis of our study showed that, for cryoablation, overweight/obesity was not a significant risk for AF recurrence. Our results showed a positive correlation between LA dimension and BMI, which may explain the reason why differences in ablation procedures modified the relationship between BMI and the rate of AF recurrence. To further test the modifying effects of ablation procedures on the relationship between BMI and the post-ablation AF recurrence, it would be useful to study the electrical and histological changes of the LA after ablation procedures using catheter-based potential mapping and cardiac MRI, respectively.
Study Limitations
Our study has some limitations that should be noted. First, this is a single-centre, non-randomised study; thus, large-scale randomised studies are needed to confirm our findings. Second, intermittent rhythm monitoring might underestimate the actual AF recurrence rate. As in previous studies, the rate of AF detection by device interrogation was also significantly higher in this study.29 Thus, reassessment with other modalities may be necessary. Third, LA diameter rather than LA volume was used in our analysis. Fourth, this study cannot make a clear conclusive remark about whether obesity is a confounder or an effect modifier of success rates of ablation from the perspective of persistent versus paroxysmal AF versus LA dimension. Finally, this study performed a race-specific BMI assessment. Providência et al. have shown comparable efficacy between cryoablation and radiofrequency ablation in patients with a BMI >30 kg/m2 in Europe.18 Although a study stratified by the same BMI would be needed, this was difficult in this study because most patients had a BMI <30 kg/m2 (Figure 3B). However, the severity of obesity is different between Asian and western people, and information about the effects of ablation on AF recurrence, specifically in Asian patients with a BMI >25 kg/m2, may be important. Research using a more accurate method to assess obesity is desirable.
Conclusion
We found that AF recurrence in Japanese patients undergoing radiofrequency ablation, but not in those undergoing cryoablation, increased with BMI, especially in patients with a BMI of ≥25 kg/m2. Thus, cryoablation may be superior to radiofrequency ablation in Japanese patients who are classified as overweight/obese.
Cick here to view Supplementary Material.
Clinical Perspective
- Multivariate logistic regression analysis identified larger left atrial dimension and radiofrequency ablation as independent predictors of AF recurrence in patients classified as overweight/obese.
- Higher BMI was significantly associated with AF recurrence in the radiofrequency ablation group, but not in the cryoablation group.
- Left atrial dimension was positively correlated with BMI and was larger in the persistent AF group than in the paroxysmal AF group.