The COMBO stent (OrbusNeich Medical) is a novel stent comprising an abluminal coating of a biodegradable polymer for eluting sirolimus and a luminal prohealing anti-CD34+ antibody layer for capturing circulating endothelial progenitor cells and providing rapid endothelialisation. This novel technology may shorten the duration of dual antiplatelet therapy (DAPT) after stenting. Evaluation of clinical outcomes after COMBO stent implantation in randomised trials and large, prospective, multicentre registries showed low clinical event rates and the noninferiority of the COMBO stent versus ‘first in class’, second-generation and newer-generation drug-eluting stents (DES).1–5
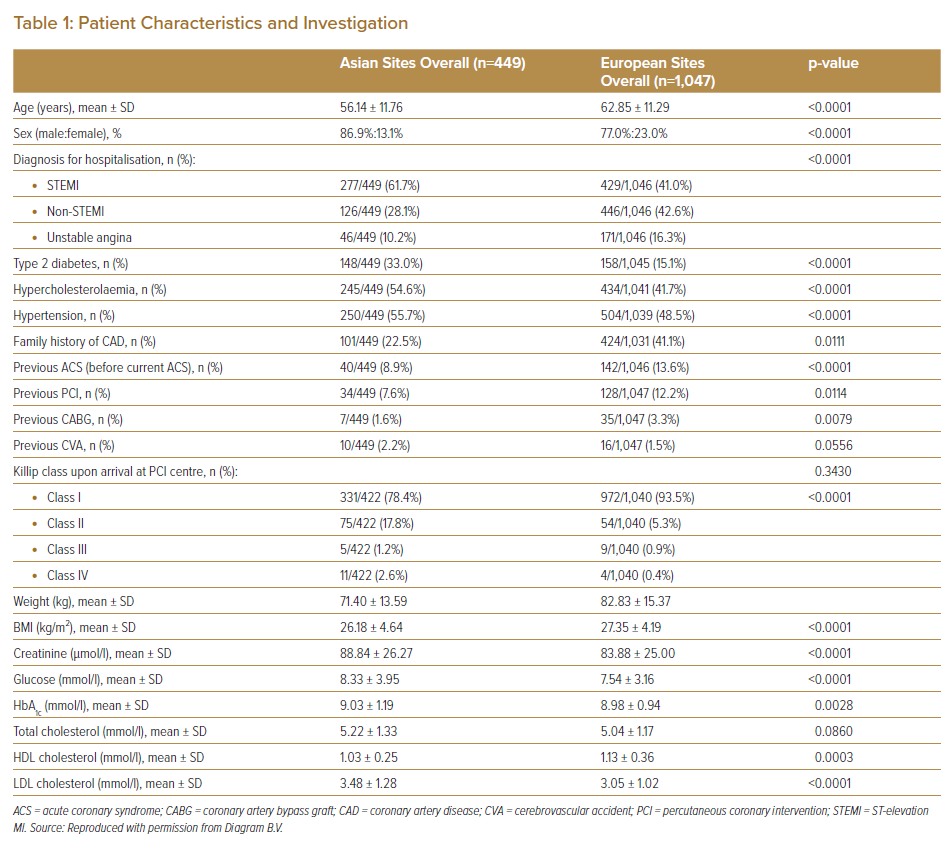
The demographics and patterns of diseases differ between patients from the Asia-Pacific region and Europe. The results of coronary angiography reveal that Asia-Pacific patients have relatively smaller coronary arteries but longer lesion length than European patients.6 In addition, more recent antithrombotic treatment trials have shown that Asia-Pacific patients have a higher risk of bleeding (particularly gastrointestinal bleeding and haemorrhagic stroke) than European patients.7–9
With the advent of newer-generation DES, real-world clinical data comparing patient profile, procedure details and clinical outcomes between Asian and European patients remain scarce. Thus, this study aimed to assess these regional differences in the era of contemporary percutaneous coronary intervention (PCI) practice.
Methods
The REDUCE (Short-term Dual Anti Platelet Therapy in Patients With ACS Treated With the COMBO Dual-therapy Stent) trial (NCT02118870) is an investigator-initiated prospective, multicentre, randomised trial involving patients with acute coronary syndrome (ACS) after treatment with COMBO stent randomised to either 3 or 12 months of DAPT. The primary study endpoint was a composite of all-cause mortality, MI, stent thrombosis, stroke, target-vessel revascularisation (TVR) and bleeding at 12 months. The data management team of Diagram B.V. (Zwolle, the Netherlands), a contract research organisation specialising in tailored cardiovascular research, provided the data for this post-hoc analysis. These unpublished data (not included in the main paper of the REDUCE trial and not published elsewhere) were obtained from the subgroup analysis report of Asian sites dated 12 February 2020 using Diagram B.V. (Supplementary Material). The main study was designed to enrol 1,500 patients with ACS from 36 investigational sites in Europe and Asia. The Asian sites were Malaysia (252), Singapore (81), Indonesia (60) and Hong Kong (57), with a total of 450 patients, and the European sites included the Netherlands (589), Belgium (254), Italy (150), Germany (31), Poland (16) and Hungary (10), with a total of 1,050 patients.
The REDUCE protocol, which describes the study design and randomisation, ethics approval, treatment and follow-up procedures, study endpoints and statistical consideration, was published in the American Heart Journal, with the trial results published in EuroIntervention in 2019.5,10 The main inclusion criteria were patients with a diagnosis of ACS (ST-elevation MI [STEMI], non-STEMI [NSTEMI], or unstable angina), successful COMBO stent implantation (thrombolysis in MI [TIMI] III flow with residual stenosis of <20% based on visual estimation) and no clinical adverse events during hospitalisation (death, stent thrombosis, stroke, TVR, bleeding with Bleeding Academic Research Consortium [BARC] classification II, III and V). The main exclusion criteria were patients with cardiogenic shock, recent major bleeding complications or contraindication to DAPT, oral anticoagulation requirement, bleeding diathesis history, stroke or transient ischaemic attack history within the last 6 months, gastrointestinal or genitourinary bleeding history within the last 2 months and/or major surgery within 6 weeks.
Of the 1,500 eligible patients with ACS who underwent successful COMBO stent implantation, four revoked their informed consent soon after randomisation, leaving 1,496 patients for analysis. Of these, 449 (30%) were Asian patients and 1,047 (70%) were European patients. The overall patient data were compared between the two sites.
Patient and Public Involvement Statement
The subgroup analysis of this study using patient data from the main REDUCE trial did not involve the patients and the public.
Statistical Methods
Cumulative event rates were estimated using the Kaplan–Meier method and compared using the log-rank test. The incidence of events was compared using chi-square or Fisher’s exact test. Student’s t-test was used for normally distributed variables and Mann–Whitney U-test for non-normally distributed variables. Categorical variables were summarised using number and percentages and compared using two-sided chi-square tests or Fisher’s exact test, as applicable.
Results
At Asian sites, the 3-month DAPT cohort showed a higher proportion of hypercholesterolaemia cases than the 12-month DAPT cohort (59.6% versus 49.6%; p=0.0319). The number of cases of infarct-related vessel (IRV) was also significantly higher in the 3-month cohort than in the 12-month cohort (p=0.0078), with IRV in the left anterior descending (LAD) artery contributing to the largest difference (67.7% versus 56.6%). At European sites, the 12-month DAPT cohort comprised more women compared with the 3-month cohort (27.0% versus 19.1%; p=0.0026). Other patient characteristics, procedure details and discharge data showed no significant differences between these cohorts for both Asian and European sites.
Moreover, the Asian cohort was younger and showed a higher prevalence of conventional cardiovascular risk factors (type 2 diabetes, hypercholesterolaemia and hypertension) and STEMI, but a lower prevalence of known family history of coronary artery disease (CAD), ACS, PCI and coronary artery bypass graft (CABG) surgery than the European cohort. Furthermore, Asian patients had higher Killip classification levels upon arrival at the PCI centre as well as lower weight and BMI, higher serum creatinine and glucose levels and worse lipid profiles than European patients (Table 1).
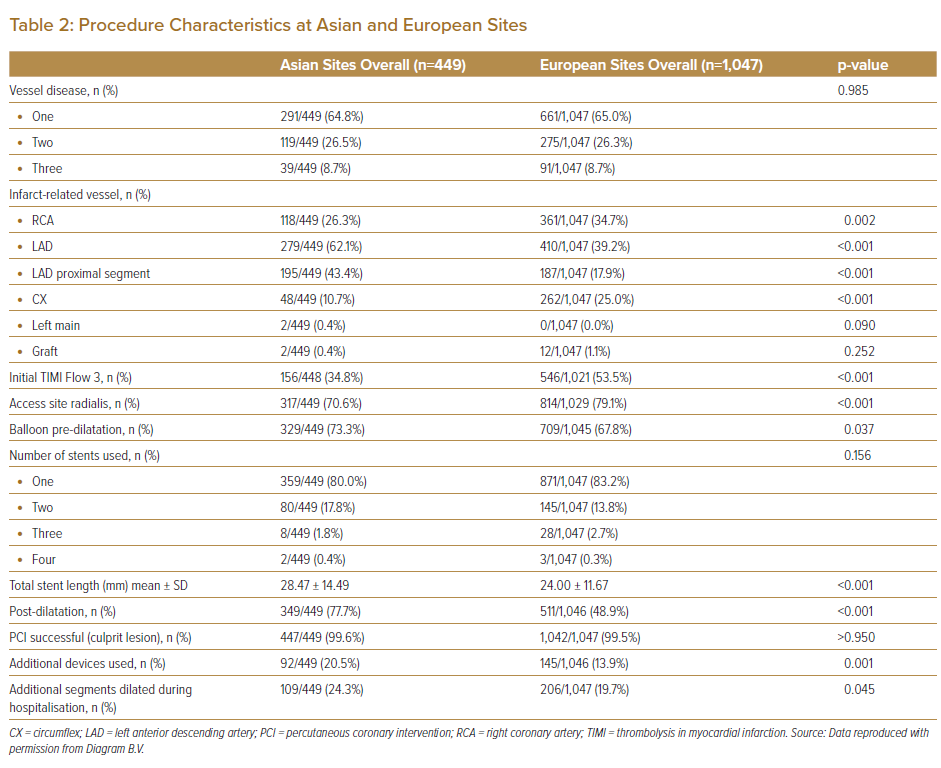
In terms of the procedure, Asian patients had a notably higher number of IRV cases in the LAD artery and at the proximal segment of LAD than European patients. Furthermore, these Asian patients had lower rates of initial TIMI III flow and radial site access and a higher incidence of balloon pre-dilatation and post-dilatation before and after stent deployment. Nonetheless, the PCI success rate (approximately 100%) and the number and length of stents used were similar between the two geographical groups. The incidence of using additional devices, such as those for embolic protection and thrombosuction, was more frequent among Asian patients than among European patients (Table 2).
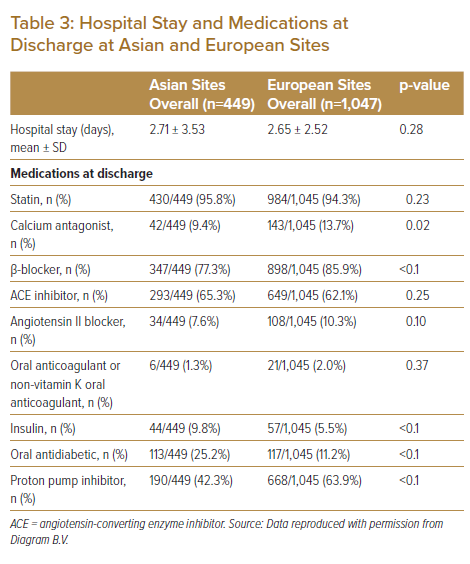
The length of hospital stay was similar between the two geographical groups. On discharge, fewer Asian than European patients were administered with calcium antagonists, β-blockers, oral anticoagulants and proton pump inhibitors. Asian patients were twice as likely to be receiving diabetic medications (insulin or oral antidiabetic medications) than the European patients (Table 3).
Outcomes
Figure 1 shows data regarding the number of patients available for the outcome analysis at various time intervals.
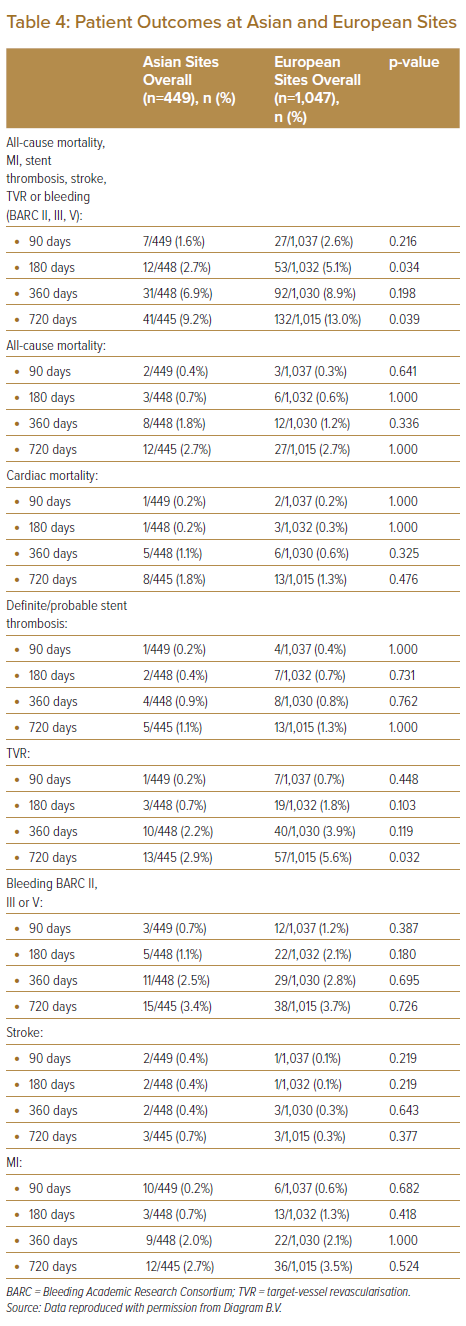
The composite primary endpoint indices for all-cause mortality, MI, stent thrombosis, stroke, TVR and bleeding (BARC II, III, or V) were lower among Asian patients than among European patients at 90, 180, 360 and 720 days (1.3% versus 2.6%, 2.7% versus 5.1%, 6.9% versus 8.9% and 9.2% versus 13.0%, respectively). This decrement among Asian patients is attributed to the lower number of cases of TVR (0.2% versus 0.7%, 0.7% versus 1.8%, 2.2% versus 3.9% and 2.9% versus 5.6%), bleeding (BARC II, III, or V) (0.7% versus 1.2%, 1.1% versus 2.1%, 2.5% versus 2.8% and 3.4% versus 3.7%) and MI (0.2% versus 0.6%, 0.7% versus 1.3%, 2.0% versus 2.1% and 2.7% versus 3.5%) at 90, 180, 360 and 720 days, respectively. However, no difference was found in all-cause mortality and cardiac mortality between Asian and European patients. Although PCI was performed in patients with ACS, the incidence of stent thrombosis was extremely low in both groups (Table 4).
Discussion
Using the data of the main REDUCE trial, this study showed that among patients with ACS treated with the COMBO stent, the primary outcomes (all-cause mortality, MI, stent thrombosis, stroke, TVR and bleeding [BARC II, III, or V]) were not different between the 3- and 12-month DAPT cohorts during follow-up at 1 and 2 years.5 This analysis provides a unique opportunity to compare the differences in the clinical characteristics, procedures and outcomes of patients with ACS in the era of new-generation DES. Moreover, it provides an opportunity for comparing contemporary practices between the Asian sites (Malaysia, Singapore, Hong Kong and Indonesia), which share various similarities regarding ethnicity, patient profile, cultural and psychosocial factors and PCI procedures, and European sites (the Netherlands, Belgium, Italy, Germany, Poland and Hungary), which also share these similarities.
Our data revealed that the Asian cohort was younger, comprised a higher number of women, had a higher prevalence of cardiovascular risk factors, a lower prevalence of family history of CAD and a higher incidence of STEMI at presentation than the European cohort. Furthermore, Asian patients showed lower prevalence of history of ACS and revascularisation (PCI and CABG) than European patients. In addition, Asian patients showed a higher Killip classification level at presentation, with higher serum creatinine and blood glucose levels and worse lipid profiles than European patients. The incidence of the culprit segment being located at the proximal LAD was also higher among Asian patients than among European patients. Asian patients also had a lower rate of initial TIMI III flow before PCI. In terms of procedure, Asian patients had a lower rate of radial site access but had higher rates of balloon pre-dilatation and post-dilatation and the use of additional devices. This finding reflects the differences in practice between Asian and European PCI operators. Nevertheless, the procedural success rates and length of hospital stay were similar between the two geographical groups. At discharge, Asian patients were administered with fewer prescriptions of β-blockers, oral anticoagulants and proton pump inhibitors and were twice as likely to be receiving diabetic medication. The primary endpoint indices at 90, 180, 360 and 720 days were also lower among Asian patients than among European patients, which can be attributed to lower TVR, bleeding (BARC II, III or V) and MI cases among Asian patients. However, all-cause mortality, cardiac mortality and stent thrombosis were similar between the two geographical cohorts.
In the e-HEALING worldwide registry substudy by Klomp M et al., differences in cardiovascular risk factors and clinical outcomes were evaluated between Western European and Southeast Asian patients treated with the Genous Bio-engineered R stent (OrbusNeich Medical Technologies).11 The primary study outcome was target-vessel failure (TVF), which is the composite of cardiac death or MI and TVR, at 12-month follow-up. Of 3,604 eligible patients, 2,873 (80%) were European and 731 (20%) were Southeast Asian. The findings are similar to those of our study, which used patient data from the REDUCE trial. For instance, Southeast Asian patients were younger compared with European patients (mean age 57.4 ± 9.9 versus 65.9 ± 11.2 years, p<0.01), with fewer women (15% versus 25%, p<0.01). Southeast Asian patients also had a higher incidence of type 2 diabetes (36.0% versus 24%, p<0.01) and a lower prevalence of MI or revascularisation history. In comparison with European patients, their average lesion was longer but their vessel diameter was smaller. At 12-month follow-up, the primary endpoint of TVF was found in 11.4% of European patients and only 5.6% of Southeast Asian patients (p<0.01). This previous study concluded that differences exist in patient clinical profiles and outcomes between the European and Southeast Asian patients who have undergone PCI.11 This suggests that reports from studies worldwide should include both overall and regional subgroup outcomes.
A more recent study by Krackhardt et al.12 assessed the regional and ethnic differences in an unselected patient population treated with newer-generation DES (polymer-free sirolimus-eluting stents) in Asia and Europe. Of the 7,190 eligible patients, 3,186 (44.0%), 2,317 (32.0%), 1,274 (17.6%) and 413 (5.7%) were from the Mediterranean region, Central Europe, South Korea and Malaysia, respectively.
Their findings also showed differences in all cardiovascular risk factors across the regions. Although Central European patients were the oldest (68.0 ± 10.9 years) and Malaysian patients were the youngest (59.7 ± 10.9 years), the incidence of type 2 diabetes was markedly different, with rates of 48.7% in Malaysia and 40.1% in Central Europe. The Malaysian subgroup also showed longer lengths of lesions and stents than the European subgroup (22.8 ± 8.6 versus 17.4 ± 9.8 mm and 23.7 ± 7.1 versus 20.7 ± 10.3 mm, respectively). The pre-dilatation rate at the Asian sites (South Korea, 94.5%; Malaysia, 86.2%) was higher than that in Central Europe (58.9%). This difference is attributed to smaller vessel diameter and longer lesion length among Asian patients. In the combined ACS/elective CAD cohorts, the primary endpoint target-lesion revascularisation (TLR) was borderline significant (p=0.046) across regions, with 2.4%, 2.5%, 1.5% and 0.8% in the Mediterranean region, Central Europe, South Korea and Malaysia, respectively. The accumulated TLR rates for both ACS and stable CAD were significantly lower in Malaysia than in Central Europe (ACS, 1.2% versus 3.1%; stable CAD, 0.8% versus 2.1%).
Moreover, the ASPECT collaboration examined the variation in clinical characteristics and outcomes between patients undergoing PCI across the Asia-Pacific region.13 Of a total of 52,608 cases from 27 hospitals, 48.4% were from Malaysia, 7.7% from Singapore, 4.8% from Hong Kong and 39.1% from Australia. Asian patients were younger and comprised fewer women than Australian patients. They also had higher incidence rates of diabetes, dyslipidaemia and renal failure but showed a lower prevalence of family history of ACS. However, the procedural success rates were similar across the region (>95%).
In summary, available data from the registries and randomised controlled trials from the eras of bare-metal stents and contemporary DES show that patient clinical profiles, procedure findings and outcomes – particularly the TVR rate – are significantly different between Asian and European patients, which is consistent with the findings of this study. These differences are important factors that should be considered for developing risk-adjustment models to enable meaningful outcome comparisons and for establishing benchmarks for the site to aid in providing quality care to patients.
These differences may have been caused by several factors, including sociocultural differences between the two populations, different risk factors in different populations and potential genetic influences. The difference in the PCI practices between the two sites may also contribute to the different outcomes between the two populations. The Asian patients seem to have higher rates of using pre- and post-dilatational balloons and additional devices during the PCI. However, these potential contributors to such differences still require further in-depth studies to solve this issue.
Study Limitations
This study has several limitations. It is a subgroup analysis with its inherent limitations in data interpretation. The sample size was not powered to determine the difference in the outcomes between Asian and European patients. Furthermore, the overall patient data in the 3- and 12-month DAPT cohorts for the two geographical sites were consolidated to simplify the comparison; even though most of the mentioned parameters were similar, minor differences were found between the two cohorts. Finally, this study only included patients randomised to the REDUCE trial; thus, the result may not be applicable to the general population.
Conclusion
In the REDUCE trial, patient clinical characteristics, procedures and outcomes were not significantly different between Asian and European patients, given that all p-values were not significant on univariate analysis. However, Asian patients tended to have lower primary endpoint indices for all-cause mortality, MI, stent thrombosis, stroke, TVR and bleeding (BARC II, III or V) for up to 2 years, despite having higher incidence of cardiovascular risk factors, STEMI and the culprit lesion located at the proximal LAD artery.
Click here to view Supplementary Material.
Clinical Perspective
- Differences between Asian and European patients with acute coronary syndrome in the era of new-generation stents are not well understood.
- Previous studies noted differences in patient demographics, percutaneous coronary intervention practices and outcomes between Asian and European patients, which require further exploration.
- Our outcome question is whether these differences translate into poorer outcomes in Asian patients than in European patients.
- Clinical trial data of European patients should be interpreted with caution because of their possible differences from those of Asian patients.