Tricuspid valve infective endocarditis (TVIE) is strongly associated with IV drug use (IVDU); device implants also represent significant risk factors. Congenital anomalies, including ventricular septal defect (VSD), predispose patients to TVIE.1 Streptococcus mitis is a mesophilic alfa-haemolytic germ inhabiting the oro- and nasopharyngeal cavities and has been implicated in cases of culture-negative endocarditis.2
Systemic extreme activation of the coagulation cascade leading to disseminated intravascular coagulopathy may result from sepsis, trauma or malignant disease, and is a life-threatening condition that is challenging to manage, especially when the patient requires an urgent intervention.3
Case Report
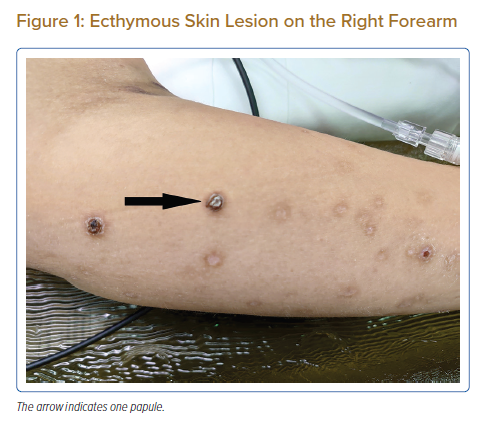
A 33-year-old woman was admitted to an external hospital with a 2-month history of increasing shortness of breath, orthopnoea and skin rash that later developed into ecthyma (Figure 1). She denied fever and any further symptoms. Her past medical history consisted of a perimembranous VSD that had not been followed up since her teenage years. There were no records of IVDU or recent dental procedures.
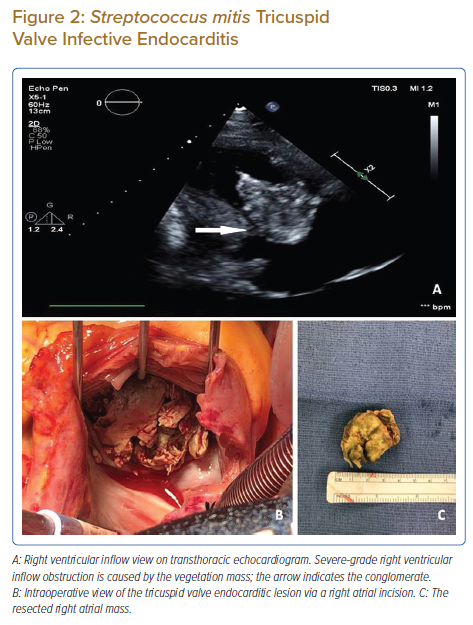
The initial echocardiogram revealed a 5.5 × 3 cm mobile mass attached to the anterior tricuspid valve leaflet (Figure 2A), resulting in moderately severe tricuspid valve stenosis with a Doppler mean trans-tricuspid pressure gradient of 4–5 mmHg and a small perimembranous VSD with a left-to-right shunt. Blood cultures were positive for S. mitis, and the patient was started on IV ampicillin. On transfer to our hospital after 1 week, the patient suddenly developed a non-bleeding type disseminated intravascular coagulopathy (DIC) with acute kidney injury and pancytopenia. The immunological origin of acute kidney failure was excluded, and she was commenced on unfractionated heparin treatment. The platelet count has not improved with the treatment; a screening test for heparin-induced thrombocytopenia (HIT) became positive. Given the size of the vegetation and her symptomatic presentation, it was decided to proceed with an urgent tricuspid valve replacement with a 31 mm biological valve (CE Perimount Magna Mitral Ease, Edwards Lifesciences) and direct VSD closure. The large, partially occlusive, cauliflower-like, friable vegetation was removed en bloc with the anterior tricuspid leaflet (Figures 2B and 2C).
Due to the positive HIT screening test and delay in obtaining a follow-up serotonin release assay, it was decided to use bivalirudin for anticoagulation during cardiopulmonary bypass. Before cannulation, a bolus of 0.75 mg/kg was administered, and 5 minutes later the activated clotting time (ACT) test was performed. During the procedure, a 1.75 mg/kg/h infusion rate was maintained. A glomerular filtration rate under 30 ml/min significantly influences the elimination of the drug. After decannulation, bivalirudin was stopped and blood products were given according to the actual clinical picture. The patient stayed in the operative theatre so long the sufficient settling of chest drainage was achieved.4
Argatroban was restarted on postoperative day 1 given that the coagulation parameters were close to the normal range. Chest drains were retained for 4 days until minimal discharge was achieved. The patient required gradual weaning of inotropes and pressors; preoperative antibiotic treatment was completed in a total of 6 weeks, stretching into the postoperative period. Dialysis was required for a period of 15 weeks postoperatively before native renal function recovered.
A pericardial effusion was noted on postoperative echocardiograms, which progressed to moderate size without signs of tamponade by postoperative day 10, and was drained percutaneously. The drained effusion was haemoserous, and the microbiological culture showed no evidence of an infection in the obtained pericardial fluid. The pericardial collection was probably multifactorial in origin, including but not limited to positive fluid balance in the context of the slowly recovering acute renal failure, postoperative systemic inflammatory response syndrome, and temporary oral anticoagulation for a 3-month period in view of the biological valve implant.
Histology of the anterior tricuspid leaflet and vegetation block showed thrombotic material without signs of acute endocarditis. Tissue stains were negative, including for acid-fast bacilli, and polymerase chain reaction (PCR) testing has not detected any further microbes. The anterior leaflet had no structural deformities that could have led to spontaneous thrombosis of the valve, and no immunological contributor to the process was discovered.
The patient was discharged after nearly 3 weeks of a gradual recovery in good functional status.
Discussion
TVIE surgeries can be performed with low operative mortality and excellent survival. Septic pulmonary embolism and failed medical therapy are more important reasons to operate than severe tricuspid valve regurgitation itself. S mitis infection results from poor dental hygiene; patients with valvular abnormalities or septal defects causing jet lesions on neighbouring endocardial structures are prone to the disease. Patients may develop mixed septic and cardiogenic circulatory collapse that requires urgent surgical handling. If untreated, it may result in a fatal outcome, and partially treated patients have a high in-hospital and post-discharge mortality.5 Patients who present with infective endocarditis skin manifestations have a higher rate of extracardiac complications. A link between new-onset ecthyma and streptococcal infections has been shown in a high percentage of patients.6 For blood culture-negative infective endocarditis, valve histopathology and PCR are useful.2
An overwhelming triggering of the coagulation system leads to a significant impact on clinical settings; sepsis may result in DIC.3 HIT represents an adverse drug reaction mediated by platelet-activating antibodies targeting complexes of heparin and platelet factor 4, and is characterised by an increased risk of thromboembolism. The American Society of Hematology recommends applying the 4Ts score rather than a traditional approach for estimating the pre-test probability of HIT. Recommendation 4.1.b for cardiosurgical patients suggests intraoperative anticoagulation with bivalirudin or heparin with perioperative plasma exchange if delaying surgery is not feasible.4
The present case highlights the challenges regarding differential diagnosis of infective and non-infective endocarditis, in which past medical history and histopathology play crucial roles. Non-infective endocarditis might be caused by immunological diseases, chemical agents, mechanical stress on the endocardium, malignancy or a hypercoagulable state.5 The present patient had an initial streptococcal blood culture positivity, and the presenting skin lesions were also diagnostic for the infective agent. The past medical history contained no records of causative factors for non-infective endocarditis; histological samples were inconclusive in this regard. The small VSD itself was not able to result in such extensive valvular lesions.
Conclusion
Streptococcus mitis can be slow-growing and may result in delayed endocarditis presentation or culture-negative endocarditis. In these cases, histopathology and PCR may assist the diagnosis. Infective endocarditis, accompanied by skin lesions, usually represents a more severe disease course. HIT and DIC further complicated the present case, which required a well-tailored multidisciplinary approach to overcome the acute critical condition.
Clinical Perspective
- Streptococcus mitis can be characterised by slow growth, leading to delayed endocarditis presentation or culture-negative endocarditis.
- Histopathology and polymerase chain reaction may facilitate the diagnosis.
- A more severe disease course usually features infective endocarditis with concomitant skin lesions.
- A case complicated by heparin-induced thrombocytopenia and disseminated intravascular coagulopathy requires a well-tailored multidisciplinary approach to overcome the acute critical condition; a surgical procedure is possible on bivalirudin anticoagulation.