Application of thrombosuction during percutaneous coronary intervention (PCI) is an adjunctive modality. It tends to be used in situations where ectatic vessels with a large thrombus burden are encountered. Conventional manual thrombosuction devices are limited by the possibility of incomplete extraction of the thrombus and the risk of thrombus migration because of the small lumen or insufficient suction force from a syringe. Newer dedicated mechanical thrombectomy devices with a wider lumen and continuous power aspiration might provide the potential for reduced time to perfusion, prevention of thrombus-related slow flow/no flow and possibly reduce the risk of embolisation of the thrombus. We present a case in which a novel coronary mechanical thrombosuction device enabled the safe, rapid and effective complete extraction of a large thrombus from an ectatic coronary artery, resulting in complete reperfusion.
Case Presentation
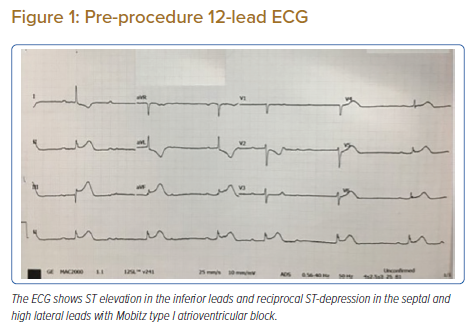
A woman aged 67 years with underlying hypertension and dyslipidaemia presented at our centre with a sudden onset of left-sided chest pain associated with diaphoresis and dizziness. Her chest pain score was 8/10 and her pulse was 40 BPM and regular. Her blood pressure was 100/70 mmHg. Her chest and other cardiovascular examinations were within normal limits. Oxygen saturation was 96% in room air. The ECG conducted at the emergency department revealed inferior ST-elevation with reciprocal ST-depression in the septal and high lateral leads, complicated by second degree atrioventricular block (Mobitz type I; Figure 1). With a diagnosis of acute inferior wall ST-elevation MI, the patient received 300 mg aspirin and 40 mg rosuvastatin in the emergency department and was taken for an emergency coronary angiogram.
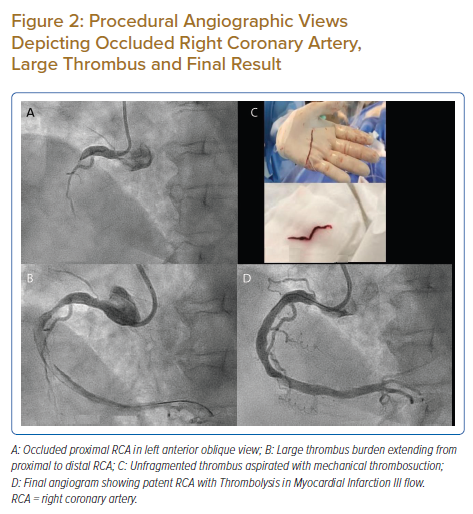
Diagnostic coronary angiography was conducted using a 5 Fr Optitorque (Terumo) catheter via the right radial artery. Left coronary artery angiogram revealed 80% stenosis in the mid left anterior descending artery, 80% stenosis in proximal ramus intermedius and minor disease in the non-dominant left circumflex artery. Her right coronary angiogram revealed a large ectatic right coronary artery (RCA) with total thrombotic occlusion at the proximal segment (Figure 2A).
The patient received 180 mg of ticagrelor and a bolus of a total of 100 IU/kg unfractionated heparin. The RCA was engaged with a 6 Fr Short Amplatz Left (SAL) guide catheter (Launcher; Medtronic). The lesion was crossed with a Sion blue coronary wire (Asahi Intecc) and pre-dilatation of the lesion was done with a 3.0 × 15 mm semi-compliant (SC) balloon. Post-ballooning, a Thrombolysis in MI (TIMI) I flow with a large thrombus extending up to distal RCA was observed (Figure 2B). Given the large thrombus burden, a loading dose of intracoronary tirofiban (25 μg/kg) was given and a maintenance dose started at 0.15 μg/kg/min. The decision was made to use mechanical thrombosuction. Thrombus aspiration was done with a 5 Fr mechanical coronary power aspiration system (Indigo System CAT RX Aspiration Catheter, Penumbra). Aspiration successfully extracted a non-fragmented massive red thrombus (Figure 2C). Thrombosuction was uneventful with TIMI III flow seen post aspiration with grade 2 myocardial blush. The culprit lesion at the proximal RCA had significant stenosis with potential plaque rupture. The lesion was further predilated with a 3.0 × 15 mm SC balloon at 16 atm. The proximal RCA was stented with a self-apposing STENTYS XPosition (STENTYS) 3.0–3.5 mm × 27 mm at 12 atm. Post-dilatation was done with a 4.0 × 15 mm non-compliant balloon at 18 atm with good angiographic results (Figure 2D). The patient’s chest pain had subsided. Rhythm had reverted to sinus rhythm and significant resolution of the ST-segment was noted. The patient had no further events during her hospital stay and was discharged with aspirin 75 mg once daily and ticagrelor 90 mg twice daily as dual antiplatelet therapy.
Discussion
Primary PCI in cases of high thrombus burden is a challenging task and is associated with an increased risk of acute and long-term complications, including stent thrombosis and death.1,2,3 A limited number of studies of aspiration thrombectomy in acute coronary syndrome have demonstrated improved myocardial perfusion demonstrated by better myocardial blush, enhanced resolution of ST-T changes and numerical reduction in in-stent thrombosis and target lesion revascularisation.4,5 Results from the single-centre TAPAS trial revealed that routine thrombus aspiration resulted in improved myocardial reperfusion compared with conventional PCI, showing significant reduction in long-term mortality during the 1-year follow-up.6,7 However, TASTE and TOTAL, the major multicentre randomised controlled clinical trials on the efficiency of routine manual thrombosuction, revealed no mortality benefit and further concerns about the increased risk of stroke emerged.8,9 A bailout treatment strategy of thrombus aspiration was used in 7.1% of those cases when conventional PCI failed and a favourable outcome was noted with thrombectomy in the subgroup analysis of cases with high thrombus load.8 Furthermore, a randomised, prospective large study on the usage of abciximab infusion and aspiration thrombectomy showed that in a large anterior wall MI, thrombus aspiration resulted in a lower rate of new-onset heart failure and hospitalisation.10
Manual aspiration thrombectomy carries a risk of thromboembolism either because of technical inadequacy or as a result of proximal clot migration and embolism because of inadequate suction pressure. Theoretically, devices with a larger lumen and sustained power aspiration throughout the procedure may offer an alternative approach to thrombosuction. These devices could limit distal embolisation as well as systemic embolisation by extracting unfragmented larger clots. Such power thrombectomy devices are an established technique in the neurovascular field for thrombus extraction and are a recommended modality. Recently, dedicated coronary mechanical thrombectomy devices have become available. In our case, we used a mechanical coronary power aspiration system (Indigo System CAT RX Aspiration Catheter), which is a 5 Fr, large-lumen continuous vacuum source power aspiration catheter system. Recently, the large prospective multicentre study CHEETAH examined this device for safety and performance, with results presented at Transcatheter Cardiovascular Therapeutics 2021. The study successfully met the primary endpoints, with low major adverse cardiac events rate and demonstrated high rates of thrombus removal, flow restoration and myocardial perfusion normalisation. TIMI flow grade 3 was achieved in 97.5% of patients, reduction of TIMI grade thrombus to zero was achieved post procedure and enhanced myocardial blush of 99.8% was reported in the study with no device related serious adverse events noted.11 In our experience, the device was technically simple to use and was able to efficiently extract a large thrombus in a single run. No adverse haemodynamic changes were noted during thrombosuction and procedure time was remarkably reduced.
Conclusion
In the setting of ectatic coronaries with high thrombus burden, mechanical thrombosuction with power aspiration might result in quicker perfusion, limited slow flow/no flow and improved immediate outcomes with, possibly, no device-related stroke. Further experience and studies with these devices are required to evaluate the efficacy and safety of mechanical thrombosuction in coronary interventions.
Clinical Perspective
- Thrombosuction during primary percutaneous coronary intervention is frequently practised to achieve good immediate reperfusion when thrombus-laden coronaries are encountered.
- Manual thrombosuction has a risk of possible thromboembolism owing to a smaller bore and insufficient suction force as the syringe fills with blood and special precautions with guiding-catheter handling are needed.
- The mechanical thrombosuction device described here overcame the limitations of manual thrombosuction because of a larger bore and sustained suction force throughout the procedure.
- This modality might assist in faster reperfusion by being able to efficiently extract larger unfragmented clots thereby avoiding risk the of no-flow and slow-flow phenomena attributed to distal thrombus migration post-ballooning.