Despite the availability of various treatment strategies, managing acute decompensated heart failure (ADHF) remains a challenge. Diuretics remain the mainstay of treatment in patients with ADHF. However, diuretic resistance (DR) can commonly occur following chronic diuretic use, leading to difficulty in diuresis.
Here we report our experience in successfully using hypertonic saline for hyperdiuresis in a critically ill patient with ADHF and severe congestion not amenable to conventional sequential nephron blockade strategies and haemodialysis.
Case Report
A 59-year-old man presented to our centre with worsening dyspnoea over a period of a day, associated with bilateral lower limb swelling. He was known to have diabetes, hypertension, dyslipidaemia, AF and Stage 3A chronic kidney disease with a baseline estimated glomerular filtration rate of 54 ml/min/1.73 m2. Vital signs on arrival included blood pressure 61/13 mmHg, requiring IV infusions of both noradrenaline and dobutamine, pulse rate 126 BPM, respiratory rate 30 breaths/min, temperature 36.5°C and oxygen saturation 88% on room air, requiring supplemental oxygen. Clinical examination revealed bilateral crepitations up to the mid-zone with evidence of gallop rhythm on auscultation of the precordium. The patient’s N-terminal pro B-type natriuretic peptide (NT-proBNP) concentration on admission was >35,000 ng/l (normal <125 ng/l).
Bedside echocardiography demonstrated severe left ventricular (LV) systolic dysfunction, suggested by an LV ejection fraction of 20–25% and LV outflow tract velocity time integral measuring 7 cm. There was evident akinesia in various regions, including the apical cap and basal to mid-lateral, anterior, posterior and anteroseptal walls. Moderate mitral regurgitation was also seen, most likely functional in nature. Lung ultrasound demonstrated B-lines in both lung fields, alongside bilateral pleural effusion.
The patient was admitted to the coronary care unit for closer monitoring and was prescribed boluses of IV frusemide, followed by IV infusion of frusemide (Figure 1). Initially, there was adequate response in diuresis, based on a urine output of 100–150 ml/h being produced in the first 96 hours. However, the patient’s urine output started to decrease soon after the 96 hours (approximately 10–20 ml/h) despite being on a frusemide infusion of 15 mg/h. The following 24 hours remained challenging as trials of sequential nephron blockade using hydrochlorothiazide and spironolactone were unsuccessful. Furthermore, following a nephrology consult, a trial of ultrafiltration was prematurely stopped due to an episode of AF with rapid ventricular response associated with haemodynamic instability. The patient’s ventilation requirement had also become more challenging, requiring non-invasive, bilevel positive airway pressure ventilation.
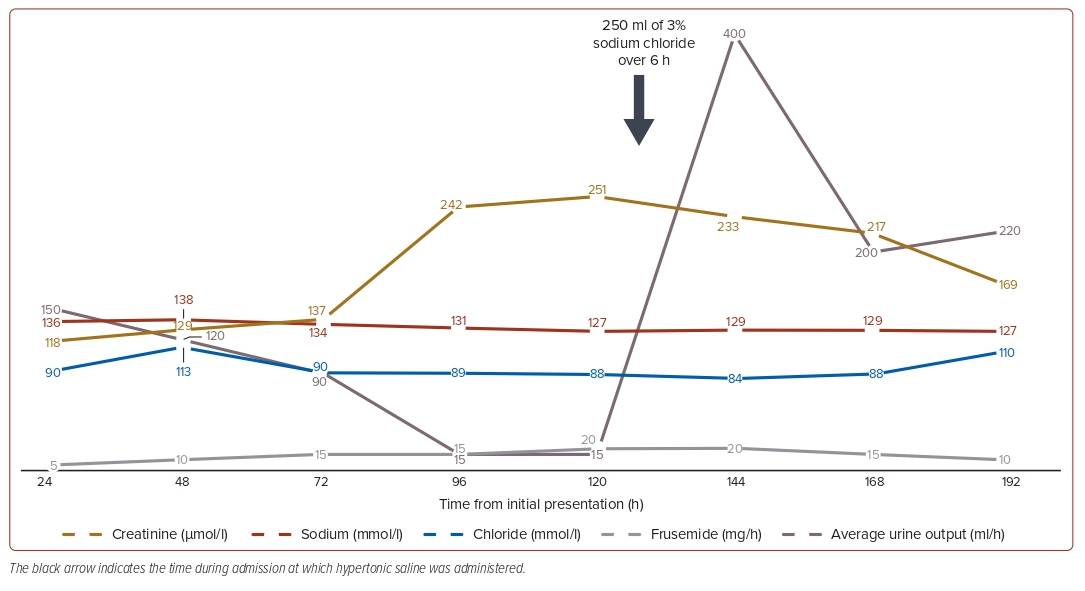
Following a review of the patient’s clinical status and biochemical parameters, a decision was made to attempt hyperdiuresis using 3% sodium chloride infusion (i.e. hypertonic saline). The treatment was administered following detailed discussion with the patient and his relatives, highlighting how the use of hypertonic saline was neither conventional nor guideline recommended, although evidence exists for its use; formal consent was obtained prior to initiation of hypertonic saline. Approximately 250 ml hypertonic saline was administered over 6 hours alongside infusion of 20 mg/h frusemide (Figure 1). There was also close monitoring of urine output and Glasgow Coma Scale, as well as serum sodium concentrations.
Within 2 hours of initiation, there were marked improvements in clinical haemodynamics and urine output, which averaged approximately 100–200 ml/h, persisting even beyond completion of hypertonic saline infusion. Within 48 hours of starting hypertonic saline, the patient improved clinically and was able to be weaned off inotrope and vasopressor support, with eventual discharge after a 9-day inpatient stay and commencement of guideline-directed medical therapy for heart failure with reduced ejection fraction.
Discussion
There is no exact definition of DR, although a common description would encompass ‘failure to decongest despite adequate and escalating doses of diuretics’.1 Between 20% and 50% of patients are deemed ‘DR’ during their hospitalisation, with DR associated with worsening heart failure, prolonged hospital admission and increased mortality.1 Furthermore, in ADHF, the exact amount of loop diuretic reaching the renal tubules often varies between individuals, compounded further by pre-existing liver and renal disease, which contribute to DR.1 Although, sequential nephron blockade has been proposed as a method to overcome DR, success varies depending on the agents used and the severity of ADHF.2
Traditionally, it was thought that high sodium intake leads to DR, predominantly in the prenephron stage.3 In addition, sodium restriction in patients admitted for ADHF is often implemented clinically, despite little evidence to support such practice. However, the recent SODIUM-HF trial has shown little to no benefit of restricting sodium intake.4 In fact, sodium restriction has been linked to heightened sodium avidity signals, leading to neurohormonal activation and potentially worsening heart failure.4
Several studies have demonstrated the utility of hypertonic saline in ADHF to varying degrees.5–7 Early studies often involved low-volume intermittent administration of sodium chloride (at different concentrations depending on serum sodium concentrations), coupled with high-dose loop diuretics to look for effective diuresis.4,5 Methods of administration have varied considerably between studies, although the evidence supporting bolus administration over infusion has since been largely disproved.8 In addition, the recent OSPREY-AHF trial has shown no benefit of modulating the sodium chloride intake of patients using an oral preparation of sodium chloride.3
The definitive mechanism as to how hypertonic saline exerts its beneficial effects remains unknown, although several hypotheses have been proposed. Because most patients in an ADHF state have reduced renal blood flow and impaired tubular secretion, with diminished intraluminal action of loop diuretics, hypertonic saline increases the concentration of intraluminal diuretics, with subsequent increases in diuresis volume, urine sodium concentrations and urine osmolarity.6,7 Poor renal blood flow is also linked to increased tubule–glomerular feedback at the macula densa, which can be ameliorated through infusion of hypertonic saline.6,7
More recently, there has been increased attention on the role of the chloride concentration, as opposed to the sodium concentration. In fact, chloride concentration has been linked to the diuretic response, neurohormonal activation and overall prognosis in patients with ADHF following a pilot study in which a short duration of sodium-free chloride infusion improved haemoconcentration and NT-proBNP concentrations.6,9,10 A likely reason for the positive outcome seen in our patient was due to the low baseline serum chloride concentrations detected throughout his admission. Other potential mechanisms that have been alluded to include improved myocardial contractility and anti-apoptotic and anti-inflammatory actions following inhibition of neutrophil activation, although more evidence is needed to substantiate these mechanisms.7
Conclusion
DR remains a major challenge when managing patients with ADHF. In cases of refractory ADHF with limited treatment options, there may be a role for the administration of hypertonic saline. However, appropriate patient selection remains crucial, and more studies are required to better understand the underlying mechanism of action, as well as to determine selection criteria to identify patients who may best benefit from this therapy. 
Clinical Perspective
- Diuretics remain the mainstay of treatment in patients with acute heart failure.
- Diuretic resistance can commonly occur following chronic diuretic use.
- In cases of refractory decompensation, there may be a role for the administration of hypertonic saline.










